VVT Medical plans to go public on Toronto exchange
VVT Medical, a developer of minimally invasive treatments for venous insufficiency, announced that it has received conditional approval from the TSXv to list its common shares.
The Kfar Saba, Israel–based company has already secured $6.5 million in funding toward its listing and seeks to reach at least $8 million in total financing. DXI Capital Corp. has agreed to acquire all outstanding shares and has acquired a holding company that will then acquire VVT Medical to facilitate its listing in Toronto.
“This approval represents a significant milestone for VVT Medical as we accelerate our global growth strategy and drive increased value for our shareholders,” VVT Medical CEO Erez Tetro said in a news release.
“Securing a listing on the Toronto Stock Exchange will enhance our access to capital markets, empowering us to further innovate and expand our portfolio of patient-focused solutions addressing venous insufficiency — a $60 billion market currently underserved, with only about 2% of patients receiving effective treatments.”
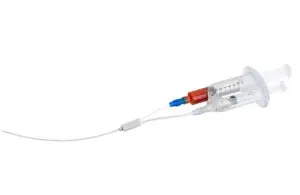
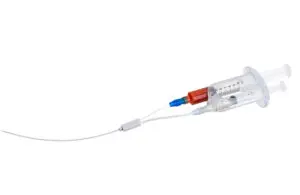
The FDA in 2023 cleared VVT Medical’s ScleroSafe platform, which delivers endovenous chemical ablation to treat superficial varicose veins. The patented coaxial, double-lumen catheter is designed for treating small peripheral veins, providing what the company describes as an efficient, non-thermal, non-tumescent treatment. VVT Medical’s portfolio also includes V-Block, which adds a permanent biomedical implant to the ScleroSafe technology and is meant to treat larger central veins.
“Combining FDA approval with strong endorsement from leading medical societies, such as the American Venous Forum (AVF), for the use of existing reimbursement codes significantly bolsters our U.S. expansion strategy,” Tetro said. “As the sole standalone company in the U.S. market with FDA approval for our novel non-thermal treatment, we are uniquely positioned.”